Zybio hs-cTnI Assay Achieves IFCC C-CB Listing: Global Witness for Excellence in Cardiac Diagnostics
August 22, 2025 Zybio News
-

December 26, 2025
DiagHub Online Academic Platform The ninth session on Microbiology Concludes Successfully – Highlights Recap
-

Nov 28, 2025
DiagHub Online Academic Platform The eighth session on Molecular Concludes Successfully – Highlights Recap
-

Nov 21, 2025
Zybio at MEDICA 2025: Comprehensive Laboratory Diagnostic Solution
The International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) has officially included Zybio's hs-cTnI assay on EXI CLIA system for its High Sensitivity Cardiac Troponin Assays List. This inclusion in the newly released IFCC C-CB Biomarkers Reference Tables witnesses the assay’s analytical excellence and clinical reliability, positioning Zybio among globally recognized IVD manufacturers meeting the highest standards in cardiac biomarker testing. This inclusion witnesses the assay’s analytical excellence and clinical reliability, Zybio’s commitment to advancing cardiac diagnostics, and our hs-cTnI empowering clinicians with the precision needed for timely, life-saving decisions.
IFCC C-CB: Global Authority Driving Standardization in Cardiac Care
The IFCC Committee on Clinical Applications of Cardiac Bio-Markers (C-CB), a definitive global body for cardiac biomarker standardization, rigorously maintained Biomarkers Reference Tables. To be included in this table, assays should meet the high-sensitivity standard as ≤10% CV at 99th percentile, ≥50% detection above LoD in healthy individuals, and cleared by major regulators (FDA, CE, NMPA).
hs-cTnI: Facilitating Cardiac Injury Detection
Cardiac troponin I (cTnI) is a regulator protein that is part of the troponin complex within cardiac myocytes, playing a critical role in myocardial contraction by modulating actin-myosin interactions in response to calcium. hs-cTnI assay quantifies very low troponin concentrations with imprecision ≤10% at the 99th-percentile upper reference limit (URL) and detects measurable values in at least 50% of healthy individuals, enabling earlier detection of injury and resolution of small deltas over short intervals. A rise and/or fall of hs-cTnI with at least one value above the 99th-percentile URL signifies acute myocardial injury. When accompanied by clinical evidence of ischemia, this also supports the diagnosis of myocardial infarction. Beyond acute coronary syndrome (ACS), detectable hs-cTnI levels provide prognostic value across cardiovascular and noncardiovascular conditions and even in asymptomatic populations, though interpretation requires clinical context given chronic elevations (e.g. CKD, older adults).
Compared with conventional troponin, hs-cTnI provides greater analytical sensitivity and precision, shortening the time from symptom onset to a detectable concentration and enabling rapid rule-in/rule-out algorithms. It outperforms CK-MB and myoglobin, which add no diagnostic value for acute myocardial injury when hs-cTn is available. Compared with hs-cTnT, hs-cTnI is similarly cardiac-specific; skeletal muscle cross-reactivity is more of a concern for cTnT, whereas cTnI elevations are cardiac in origin. Biological variation is low, supporting use of small absolute or relative deltas over 1-3 hours with assay-specific cutoffs.
| Analytical Characteristics of Zybio hs-cTnI assay | |||
| Overall | Female | Male | |
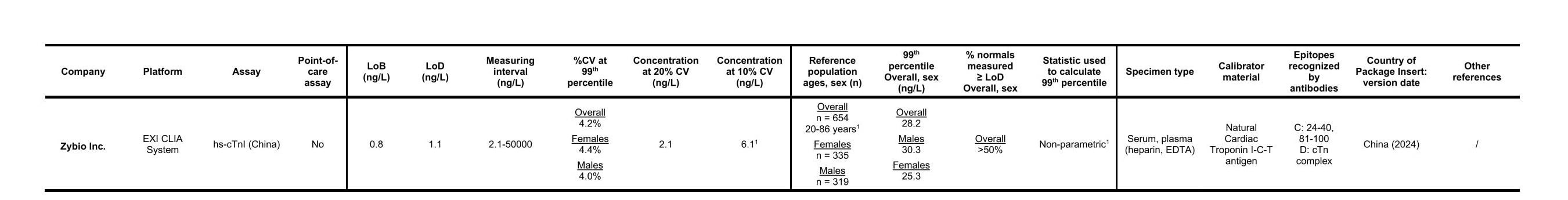
| 99th URL | 28.2 ng/L | 25.3 ng/L | 30.3 ng/L |
| 99th URL CV | 4.2% | 4.4% | 4.0% |
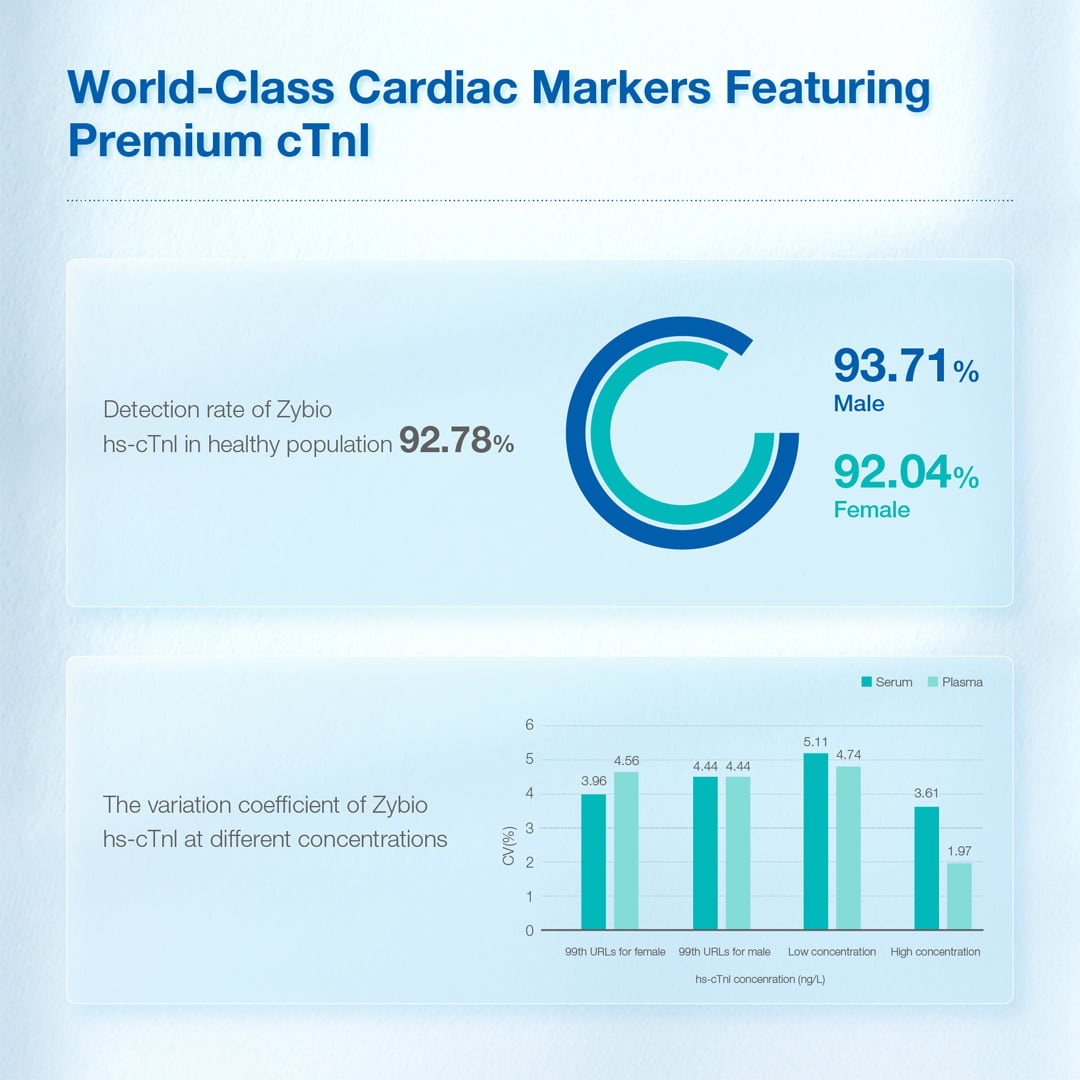
| Normals measured ≥ LoD | 92.78% | 92.04% | 93.71% |
| LoB | 0.8 ng/L | ||
| LoD | 1.1 ng/L | ||
| Duration to first result | 12.8 min | ||
Zybio hs-cTnI: Outstanding Performance & Value for Confident Clinical Decisions
Zybio’s IFCC-listed hs-cTnI assay delivers exceptional performance:
√ Wide linear range ranging 2.1-50000 ng/L, with exceptional sensitivity
√ Reliable performance with overall 4.2% CV at 99th percentile
√ 92.78% normals measured ≥ LoD
√ Reliable reference population across 20-86 years old healthy individuals with considerable number of males and females
√ Independent raw material development by Zybio, dedicated to reliable and stable quality
 |  |
→ Access the Full IFCC C-CB List HERE
Dedication to Accuracy, Zybio has been and will always be committed to improvement of total laboratory diagnostic solution and increasingly excellent products and services for partners. In this way, we aim to jointly facilitate the healthcare industry.