Positive AFB stain? Cavitary tuberculosis? “Man-machine” integration helps reveal the truth
Nov 17, 2023 Zybio News
-

December 26, 2025
DiagHub Online Academic Platform The ninth session on Microbiology Concludes Successfully – Highlights Recap
-

Nov 28, 2025
DiagHub Online Academic Platform The eighth session on Molecular Concludes Successfully – Highlights Recap
-

Nov 21, 2025
Zybio at MEDICA 2025: Comprehensive Laboratory Diagnostic Solution
By Jiang Yuhang, Qin Weichao, Li Jian, Gao Yuan, Liao Xin, Zeng Yan, Jiangjin Hospital affiliated to Chongqing University
01/ Background
In the first half of 2022, a special patient was admitted to the Respiratory Department and Tuberculosis Clinic of our hospital. The patient had received short-term treatment in the other hospital because of cough, expectoration, and fever, during which, the chest CT images of the patient showed that there were cavities in the right upper lung and middle lobe, and that the right pleura was thickened and adhered. The doctor in the other hospital highly suspected it was cavitary tuberculosis. In order to seek further diagnosis and treatment, the patient came to us for sputum smear acid-fast bacilli (AFB) staining, and at the same time, because the patient’s physical symptoms did not improve significantly, he went to the Respiratory Department for hospitalization to relieve his symptoms.
02/ Diagnosis Process
The patient sent three sputum samples for acid-fast bacilli staining, and the quality of sputum was all qualified. After acid-fast bacilli staining, the smears were examined by microscopy. All the three smears showed many acid-fast bacteria under the microscope and they were distributed in clusters. But interestingly, the acid-fast bacteria under the microscope looked obviously different from common mycobacteria. They were in a spherical form rather than rod-shape and were partially colored. Because the author had consulted the literature related to AFB stain before, the first conclusion flashing in the author's mind was that these acid-fast bacteria were “Rhodococcus equi”. However, the author knows that Rhodococcus equi, like Cryptococcus neoformans, is an opportunistic pathogen, which is usually observed in some immunocompromised patients, especially AIDS patients. Therefore, the author communicated with the doctor of the Tuberculosis Clinic Department in time, telling the doctor that the patient might be infected with Rhodococcus equi, and issued an acid-fast report with the words “acid-fast bacteria found”. According to the feedback of the Tuberculosis Clinic Department, the patient had been treated in the Respiratory Department, and his HIV antibody was positive in the initial screening, so further HIV confirmation was needed, and his CD4+ T cell count was only 31 cells/μL. At this moment, the author highly suspected that the acid-fast bacteria distributed in clusters of the sputum smear under the microscope of the patient were Rhodococcus equi. As a result, the author communicated with the patient's resident doctor again about the situation. After parallel sputum culture, the bacteria were examined by Zybio’s mass spectrometer and the result showed that they were Rhodococcus equi. (Subsequently, we cooperated with Zybio to conduct 16S rRNA molecule sequencing and the result showed that it was Rhodococcus equi). The result of the subsequent T-SPOT testing was negative, so the infection of Mycobacterium tuberculosis could be basically excluded. After the timely feedback from the author, the clinician immediately used levofloxacin, meropenem and rifampicin to treat the patient with Rhodococcus equi infection. After two weeks of treatment, the patient's symptoms improved obviously, so he was discharged from the hospital and told to continue taking oral medication after discharge. After two months of follow-up, all the infection indexes of the patient returned to normal level, and his symptoms were dramatically improved.
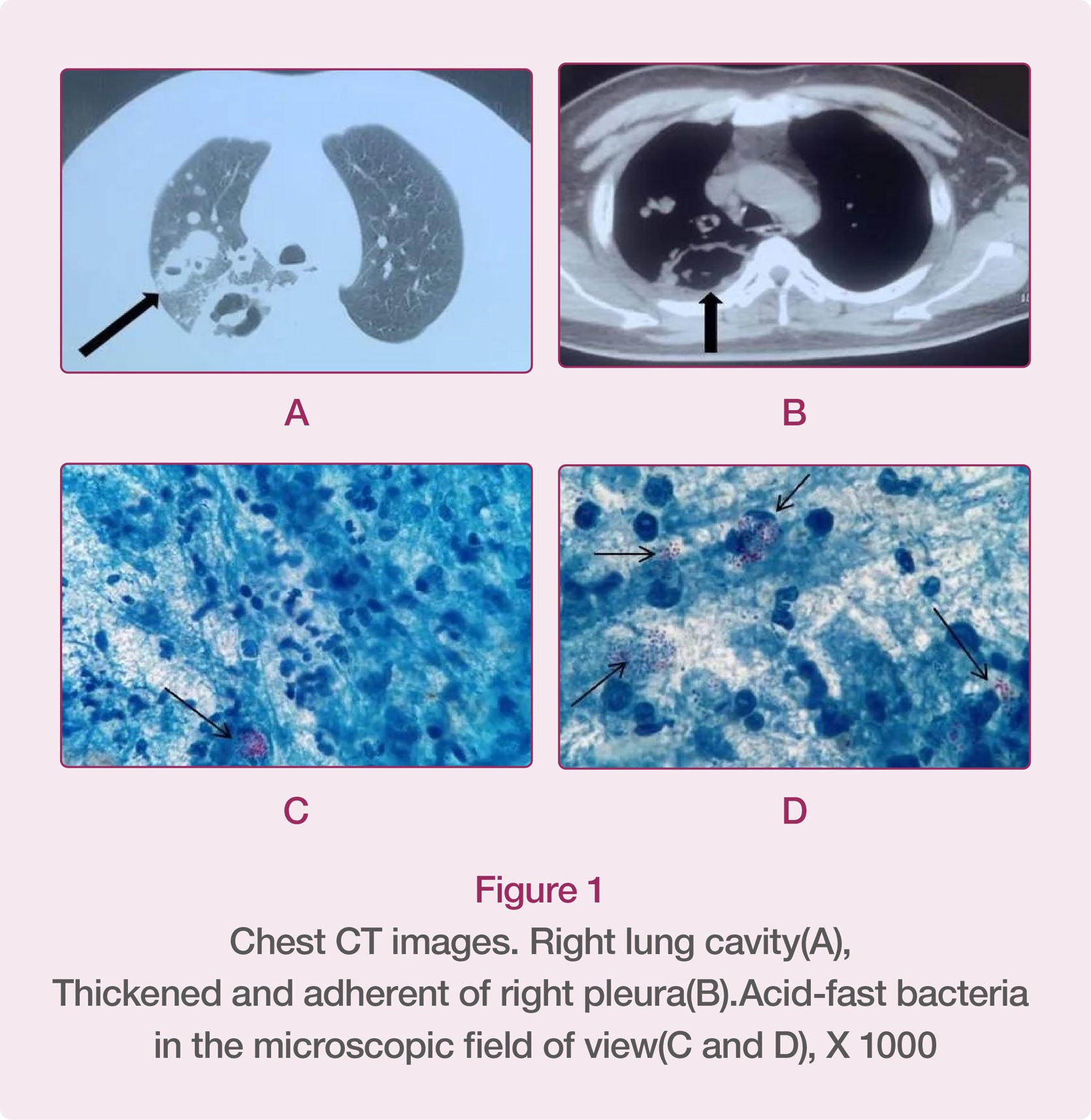
03/ Description of the Bacterium
Rhodococcus equi belongs to the genus Rhodococcus, an opportunistic pathogen, and is widely distributed in the environment (except Antarctica). The morphology of Rhodococcus equi is very interesting. It can change according to the culture environment and time. The author cultured it in an environment with 5% CO2 at 35℃ for 6h, and then the bacterium in its original area was stained with gram, showing positive under the microscope. After 24h pure culture, the bacterial colonies were light orange mucoid colonies. Currently, the gram-stained morphology of the bacterium is positive cocci. After 48h pure culture, substances similar to fungal aerial hyphae were formed around the bacterial colony, and Rhodococcus equi’s weak acid-fast bacilli staining was positive, showing partial staining. In addition, hexamine silver staining can also stain it as dark brown as fungi.
04/ Discussion
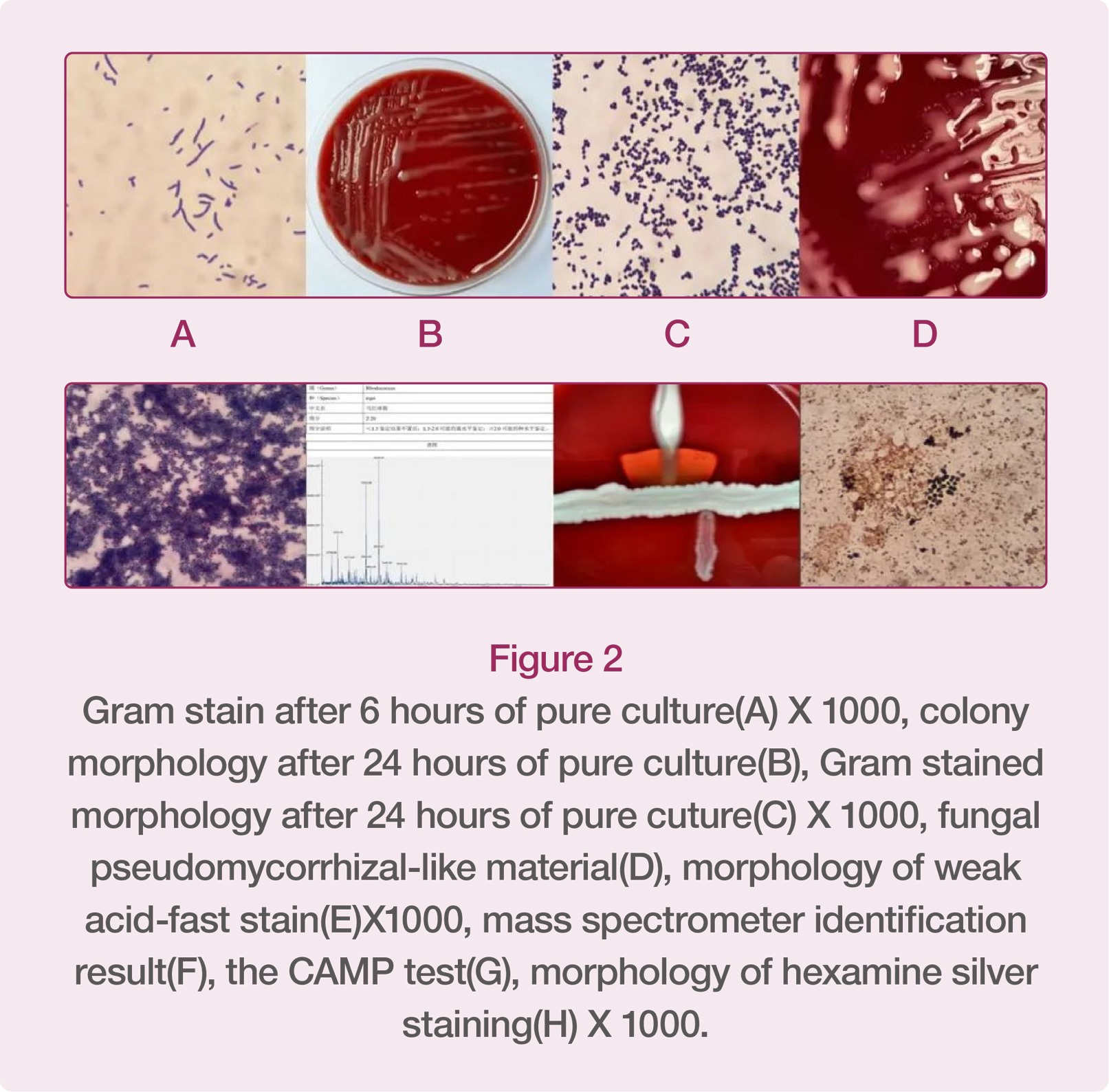
Rhodococcus equi belongs to the genus Rhodococcus. At present, there are 57 species of Rhodococcus, and Rhodococcus equi, Rhodococcus erythrorhizon and Rhodococcus rhodochrous are common in clinical medicine. Rhodococcus equi is an opportunistic pathogen widely distributed in nature, and is closely related to HIV infection. According to foreign literature, about two-thirds of the patients infected with Rhodococcus equi are also HIV-infected patients, so we should be vigilant about it in clinical practices. It is easy to misdiagnose Rhodococcus equi infection. On the one hand, (1) misdiagnosis can be caused by the accuracy of laboratory identification. The morphology of gram staining of Rhodococcus equi is related to the culture time. When bloodstream infection occurs, gram staining smear may be positive Corynebacterium when blood culture is positive in a short time. If such result is reported to the Clinical Department, the clinician may judge them as contaminated bacteria, thus missing the correct therapeutic scheme. Two cases of explosive Rhodococcus equi infection were reported abroad. Although blood cultures showed positive results many times, they were both wrongly identified as Corynebacterium, causing the clinician to judge it as contaminated bacterium. The units that use Biomerieux VITEK-2 Compact to identify drug-sensitive bacteria should pay attention to the fact that only Rhodococcus equi is included in the accompanied CBC card identification catalogue, while most units using Biomerieux instruments are only provided with GP cards. However, GP card can't be used to identify Rhodococcus equi, and it will wrongly identify it as Coxiella. If the wrong identification report is sent to a clinician, it will also cause the patient to miss the correct therapeutic scheme and treating opportunity. Therefore, mass spectrometer has unique advantages in rapid and accurate identification of bacteria. On the other hand, (2) the reason for misdiagnosis of the bacterium lies in clinical experience and judgment. Because pulmonary infection caused by Rhodococcus equi may be diffuse and pleomorphic with pulmonary cavities, it is very easy for a clinician to misdiagnose it as cavitary tuberculosis in clinical practices. The patient in this case had such clinical manifestation, which was once considered as cavitary tuberculosis when he was admitted to the hospital. In addition, Rhodococcus equi pulmonary infection can be divided into exudation stage, consolidation stage and abscess formation tage. Early lesions are often small patches of shadow with slightly higher density. If these lesions are not treated in time and effectively, they will develop into consolidation stage, forming nodular and massive consolidation lesions. At this stage, it is very similar to malignant tumor, and is often complicated with swollen hilar mediastinal lymph node. Therefore, it is easily misdiagnosed as lung cancer with hilar mediastinal lymph node metastasis.
In addition, a combination of drugs is usually recommended as the therapeutic regimen for Rhodococcus equi infection. “Sanford Guide to Antimicrobial Therapy” recommends azithromycin, levofloxacin or rifampicin as the first choice and Vancomycin or imipenem plus levofloxacin or azithromycin or rifampicin as the second choice for treating Rhodococcus equi infection. However, penicillin, cephalosporin, clindamycin, tetracycline, and compound sulfamethoxazole should be avoided (Rhodococcus equi is also resistant to the above drugs in this case). It is worth noting that although Rhodococcus equi may be sensitive to vancomycin in vitro, because Rhodococcus equi is a facultative intracelluar parasite, vancomycin can't reach the bactericidal concentration in cells, causing the activity of vancomycin to Rhodococcus equi in cells to decrease, thus ultimately leading to the failure of treatment. Therefore, drugs with sound cell penetration ability should be selected for treating Rhodococcus equi infection in clinical practices. The patient in this case was treated with levofloxacin combined with rifampicin and meropenem. After 7 days, all the indexes gradually decreased. Generally, the treatment of Rhodococcus equi infection lasts long, and requires long-term medication, and some patients still need take oral medicine after discharge. In addition, once HIV infection is confirmed, using HAART (cocktail therapy) as soon as possible can improve the prognosis of the patients suffering from HIV complicated with Rhodococcus equi infection and improve their survival rate.
Although there are more and more reports about Rhodococcus equi, many laboratories still have difficulty in identifying the bacterium or misdiagnose it, causing the patients to miss correct therapeutic schemes and appropriate treating time. This also brings great challenges to both our laboratory staff and clinicians. As laboratory staff, we should combine our own experience with various instruments to provide fast and accurate reports for clinicians. The communication between the laboratory and the clinical department is also very important. As microbiologists we should give accurate and effective information to the clinicians in time so that the patients can receive correct therapeutic schemes as soon as possible. In addition, the conventional biochemical identification method is slow, and has low accuracy in identifying rare bacteria. Mass spectrometer can be used to identify some rare bacteria rapidly and accurately, so microbiology laboratory should be equipped with mass spectrometer, which is the inevitable development trend of clinical microbiology laboratory in the future.
References
1. Lin WV, Kruse RL, Yang K, et al. Diagnosis and management of pulmonary infection due to Rhodococcus equi. Clin Microbiol Infect (2019) 25: 310-315. 2018/05/20. DOI: 10.1016/j.cmi.2018.04.033.
2. Goodfellow M. The toxonomic status of Rhodococcus equi. Vet Microbiol (1987) 14: 205–209. DOI: https://doi.org/10.1016/0378-1135(87)90106-4.
3. Weinstock DM and Brown AE. Rhodococcus equi: An emerging pathogen. Clin Infect Dis (2002) 34: 1379–1385. DOI: 10.1086/340259.
4. Tuon FF, Siciliano RF, Al-Musawi T, et al. Rhodococcus equi bacteremia with lung abscess misdiagnosed as corynebacterium: a report of 2 cases. Clinics (Sao Paulo, Brazil) (2007) 62: 795–798. 2008/01/23. DOI: 10.1590/s1807-59322007000600022.
5. Gallant JE and Ko AH. Cavitary pulmonary lesions in patients infected with Human Immunodeficiency Virus. Clin Infect Dis (1996) 22: 671–682. DOI: 10.1093/clinids/22.4.671.
6. Capdevila JA, Buján S, Gavaldá J, et al. Rhodococcus equi pneumonia in patients Infected with the Human Immunodefficiency Virus: Report of 2 cases and review of the literature. Scand J Infect Dis (1997) 29: 535–541. DOI: 10.3109/00365549709035890.
7. Savini V, Fazii P, Favaro M, et al. Tuberculosis-like pneumonias by the aerobic actinomycetes Rhodococcus, Tsukamurella and Gordonia. Microbes Infect (2012) 14: 401-410. DOI: https://doi.org/10.1016/j.micinf.2011.11.014.
8. Arya B, Hussian S and Hariharan S. Rhodococcus equi pneumonia in a renal transplant patient: a case report and review of literature. Clin Transplant (2004) 18: 748-752. 2004/11/02. DOI: 10.1111/j.1399-0012.2004.00276.x.
9. Meyer DK and Reboli A. Other Coryneform Bacteria and Rhodococci. In Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. Elsevier Inc (2014) 2: 2695-2706. DOI: 10.1016/B978-1-4557-4801-3.00207-1.
10. Topino S, Galati V, Grilli E, et al. Rhodococcus equi infection in HIV-infected individuals: case reports and review of the literature. AIDS Patient Care STD (2010) 24: 211–222. 2010/04/10. DOI: 10.1089/apc.2009.0248.
11. Kwa AL, Tam VH and Rybak MJ. Rhodococcus equi pneumonia in a patient with human immunodeficiency virus: Case report and review. Pharmacotherapy (2001) 21: 998-1002. 2001/11/23. DOI: 10.1592/phco.21.11.998.34512.
12. Lasky JA, Pulkingham N, Powers MA, et al. Rhodococcus equi causing human pulmonary infection: review of 29 cases. South Med J (1991) 84: 1217-1220. 1991/10/01. DOI: 10.1097/00007611-199110000-00014.
13. Cury JD, Harrington PT and Hosein IK. Successful medical therapy of Rhodococcus equi pneumonia in a patient with HIV infection. Chest (1992) 102: 1619-1621. 1992/11/01. DOI: 10.1378/chest.102.5.1619.
14. Tsuchida S, Umemura H and Nakayama T. Current status of matrix-assisted laser desorption/ionization-time-of-flight mass spectrometry (MALDI-TOF MS) in clinical diagnostic microbiology. Molecules (2020) 25 2020/10/22. DOI: 10.3390/molecules25204775.
15. 伍国伟,刘含秋,郭兴全,苏凌松.HIV机遇感染马红球菌病误诊原因分析[J].临床肺科杂志,2014,19(02):375-376.
* Unauthorized reprinting is prohibited.
